Biogen inventory is getting favorable rankings after profitable trial outcomes for its newest Alzheimer’s remedy
Lecanemab is Biogen’s newest try and remedy the illness after its failed rollout of one other drug final 12 months
This time, the corporate appears to have a greater shot at attaining a breakthrough
If you happen to obtained burned in final 12 months’s boom-and-bust cycle, you’re most likely hesitant to leap into Biogen (NASDAQ:) shares once more. Nevertheless, the previous two months’ rally that despatched the top off round 50% feels totally different this time round.
The present bullish pattern began after Japan-based Eisai Co (OTC:), and Biogen introduced in late September that their experimental Alzheimer’s illness drug considerably slowed the development of the mind dysfunction in a big examine.
The businesses mentioned their drug “lecanemab” diminished cognitive and practical decline by 27%, in contrast with a placebo, over 18 months in a phase-3 examine of 1,800 sufferers with early-stage Alzheimer’s. The drug was additionally linked to increased charges of mind swelling and small bleeds, although the corporate mentioned they had been not often symptomatic.
Lecanemab is the Cambridge, Massachusetts-based Biogen’s newest try and remedy Alzheimer’s after its failed rollout of one other experimental drug, “Aduhelm,” final 12 months. Regardless of getting regulatory approval from the U.S. Meals and Drug Administration, Aduhelm confronted a number of setbacks.
The company’s outdoors scientific advisers mentioned that medical trial knowledge did not clearly present that the drug labored as meant. Insurers additionally refused to pay for it, whereas the U.S. proposed tight limitations on who could be eligible to have Medicare cowl its value.
For all of 2021, Biogen had simply $3 million in Aduhelm income after spending an enormous sum on its commercialization. Consequently, its inventory plunged to a 10-year low in Could.
However this time, the corporate appears to have a greater shot at attaining a breakthrough. This week, a possible competitor, Roche (OTC:), mentioned its Alzheimer’s experimental remedy didn’t gradual cognitive decline considerably. After this failure, the one competitors left available in the market is Eli Lilly’s (NYSE:) “donanemab,” with outcomes from medical trials scheduled to be launched subsequent 12 months.
Much less competitors and the huge income alternative have prompted many analysts to improve Biogen inventory in latest weeks. In a latest Investing.com ballot, most analysts rated Biogen as a purchase.
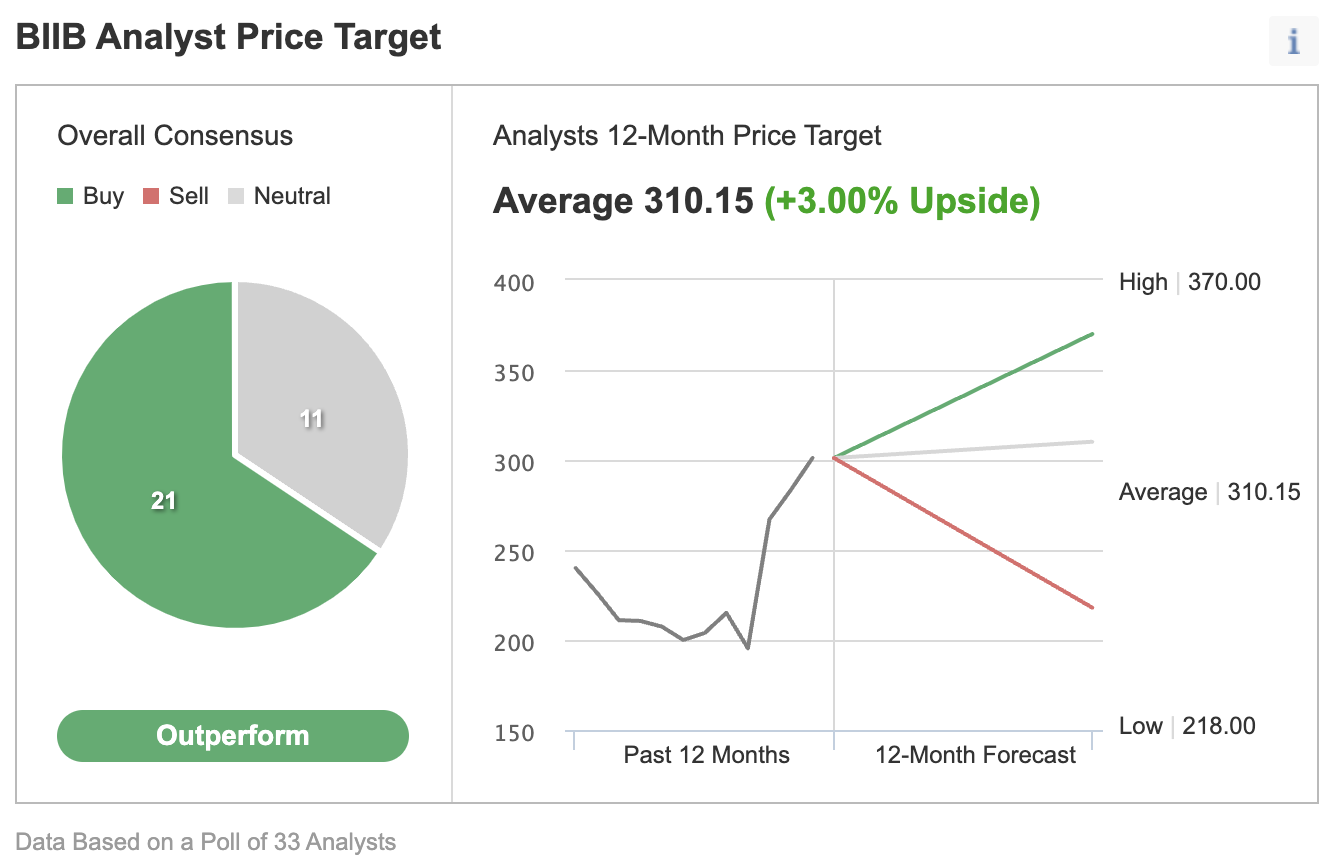
Supply: Investing.com
In a latest observe to shoppers, Goldman Sachs raised its worth goal on shares to $370, a 23% upside the place the inventory traded on Wednesday. The funding financial institution additionally raised its income estimates for the drug to $14 billion in 2035, in contrast with prior estimates of $2 billion.
GS’s observe added:
“The corporate is now poised to handle the early Alzheimer’s illness market in 2023+ following the latest topline lecanemab Ph3 (CLARITY-AD) knowledge.
Our discussions with main neurologists and administration lead us to imagine that the outcomes are clinically significant (given the profit throughout a number of endpoints, the constructive pattern over time, and the security profile).”
Biogen’s Japanese accomplice Eisai plans to submit for full approval within the U.S., European Union, and Japan within the first quarter of subsequent 12 months, and Goldman anticipates it is going to be profitable.
Regardless of this optimism, there are nonetheless loads of dangers that buyers ought to keep in mind. Whereas there’s a good chance that the FDA will approve the drug following its go-ahead of Aduhelm final 12 months, there isn’t any assure that Medicare will comply with pay for the drug, given its determination to impose restrictive reimbursement circumstances for all anti-amyloid medication—which goal an uncommon buildup of the protein in affected person’s brains.
Furthermore, even when all goes nicely, there may be little consensus on how a lot this remedy will herald income for Biogen and its Japanese accomplice. Based on a report within the Wall Avenue Journal, analyst estimates for the drug’s peak gross sales vary from a number of hundred million {dollars} to billions.
Backside Line
Biogen inventory is providing a significantly better risk-reward proposition after the success of its newest trials to deal with Alzheimer’s illness. Its 50% leap in the course of the previous two months displays the optimism that the remedy will obtain FDA approval and insurers will reimburse for the remedy.
That mentioned, Biogen inventory is for high-risk takers, given the potential for a unfavourable end result.
Disclosure: As of the time of writing, the writer would not personal shares talked about on this report. The views expressed on this article are solely the writer’s opinion and shouldn’t be taken as funding recommendation.




















